주메뉴
- About IBS 연구원소개
-
Research Centers
연구단소개
- Research Outcomes
- Mathematics
- Physics
- Center for Theoretical Physics of the Universe(Particle Theory and Cosmology Group)
- Center for Theoretical Physics of the Universe(Cosmology, Gravity and Astroparticle Physics Group)
- Center for Exotic Nuclear Studies
- Center for Artificial Low Dimensional Electronic Systems
- Center for Underground Physics
- Center for Axion and Precision Physics Research
- Center for Theoretical Physics of Complex Systems
- Center for Quantum Nanoscience
- Center for Van der Waals Quantum Solids
- Chemistry
- Life Sciences
- Earth Science
- Interdisciplinary
- Institutes
- Korea Virus Research Institute
- News Center 뉴스 센터
- Career 인재초빙
- Living in Korea IBS School-UST
- IBS School 윤리경영


주메뉴
- About IBS
-
Research Centers
- Research Outcomes
- Mathematics
- Physics
- Center for Theoretical Physics of the Universe(Particle Theory and Cosmology Group)
- Center for Theoretical Physics of the Universe(Cosmology, Gravity and Astroparticle Physics Group)
- Center for Exotic Nuclear Studies
- Center for Artificial Low Dimensional Electronic Systems
- Center for Underground Physics
- Center for Axion and Precision Physics Research
- Center for Theoretical Physics of Complex Systems
- Center for Quantum Nanoscience
- Center for Van der Waals Quantum Solids
- Chemistry
- Life Sciences
- Earth Science
- Interdisciplinary
- Institutes
- Korea Virus Research Institute
- News Center
- Career
- Living in Korea
- IBS School
News Center
|
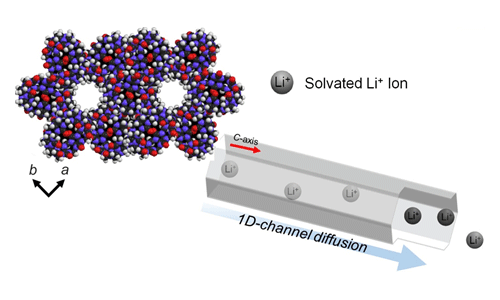
New Lithium Ion Battery isSafer, Tougher, and More Powerful - SouthKorean scientists have developed a new high-performance lithium ion batterywhich is stable at high temperatures July 3, 2015 Lithium ion batteries (LIBs) are ahuge technological advancement from lead acid batteries which have existedsince the late 1850’s. Thanks to theirlow weight, high energy density and slower loss of charge when not in use, LIBshave become the preferred choice for consumer electronics. Lithium-ion cells with cobalt cathodes holdtwice the energy of a nickel-based battery and four-times that of leadacid. Despite being a superior consumerbattery, LIBs still have some drawbacks.Current manufacturing technology is reaching the theoretical energydensity limit for LIBs and overheating leading to thermal runaway i.e. “ventingwith flame” is a serious concern. SouthKorean researchers at the Center for Self-assembly and Complexity, Institute forBasic Science (IBS), Department of Chemistry and Division of Advanced MaterialsScience at Pohang University, have created a new LIB made from a porous solidwhich greatly improves its performance as well as reducing the risks due tooverheating. Since2002 there have been over 40 recalls in the US alone due to fire or explosionrisk from LIBs used in consumer electronic devices. These types of batteries, in all of theirdifferent lithium-anode combinations, continue to be an essential part ofmodern consumer electronics despite their poor track record at hightemperatures. TheKorean team tried a totally new approach in making the batteries. According to Dr. Kimoon Kim at IBS, “we have already investigated high and highlyanisotropic [directionally dependent] proton conducting behaviors in porousCB[6] for fuel cell electrolytes. It is possible for this lithium ionconduction following porous CB[6] to be safer than existing solid lithiumelectrolyte -based organic-molecular porous-materials utilizing the simplesoaking method.” Current LIB technology relies on intercalated lithium whichfunctions well, but due to ever increasing demands from electronic devices tobe lighter and more powerful, investigation of novel electrolytes is necessaryin order.
(Above) Simple incorporation of various lithiumprecursor to porous CB[6] exhibits high lithium ion conductivities, mobilityand safer dried solid lithium electrolytes Thenew battery is built from pumpkin-shaped molecules called cucurbit[6]uril(CB[6]) which are organized in a honeycomb-like structure. The molecules have an incredibly thin 1D-channel,only averaging 7.5 Å [a single lithium ion is 0.76 Å, or .76 x 10-10 m]that runs through them. The physicalstructure of the porous CB[6] enables the lithium ions to battery to diffusemore freely than in conventional LIBs and exist without the separators found inother batteries. Intests, the porous CB[6] solid electrolytes showed impressive lithium ionconductivity. To compare this toexisting battery electrolytes, the team used a measurement of the lithiumtransference number (tLi+) which was recorded at0.7-0.8 compared to 0.2-0.5 of existing electrolytes. They also subjected the batteries to extremetemperatures of up to 373 K (99.85° C), well above the 80° C typical upper temperaturewindow for exiting LIBs. In the tests,the batteries were cycled at temperatures between 298 K and 373 K ( 24.85° C and 99.85° C) for aduration of four days and after each cycle the results showed no thermalrunaway and hardly any change in conductivity. Variousconventional liquid electrolytes can incorporate in a porous CB[6] frameworkand converted to safer solid lithium electrolytes. Additionally, electrolyte usage is not limitedto use only in LIBs, but a lithium air battery potentially feasible. What makes this new technique most exciting isthat it is a new method of preparing a solid lithium electrolyte which starts asa liquid but no post-synthetic modification or chemical treatment is needed. - By DanielKopperud Notes for editors -References JunHeuk Park,Kyungwon Suh,Md. Rumum Rohman,WooseupHwang, Minyoung Yoonand Kimoon Kim, (2015), “Solid lithiumelectrolytes based on an organic molecular porous solid”, The Royal Society ofChemistry, DOI: 10.1039/c5cc02581h -MediaContact For further information or to request mediaassistance, please contact: Mr. Shi Bo Shim, Head of Department ofCommunications, Institute for Basic Science (+82-42-878-8189; sibo@ibs.re.kr)or Ms. Sunny Kim, Department of Communications, Institute for Basic Science(+82-42-878-8135; Sunnykim@ibs.re.kr) -Aboutthe Institute for Basic Science (IBS)
|
||||
|
|
||||
| Next | |
|---|---|
| before |
- Content Manager
- Public Relations Team : Yim Ji Yeob 042-878-8173
- Last Update 2023-11-28 14:20