주메뉴
- About IBS 연구원소개
-
Research Centers
연구단소개
- Research Outcomes
- Mathematics
- Physics
- Center for Underground Physics
- Center for Theoretical Physics of the Universe (Particle Theory and Cosmology Group)
- Center for Theoretical Physics of the Universe (Cosmology, Gravity and Astroparticle Physics Group)
- Dark Matter Axion Group
- Center for Artificial Low Dimensional Electronic Systems
- Center for Quantum Nanoscience
- Center for Exotic Nuclear Studies
- Center for Van der Waals Quantum Solids
- Center for Relativistic Laser Science
- Center for Trapped Ion Quantum Science
- Chemistry
- Life Sciences
- Center for Memory and Glioscience (Cognitive Glioscience Group)
- Center for Memory and Glioscience (Learning and Memory Group)
- Center for Synaptic Brain Dysfunctions
- Center for RNA Research
- Center for Genomic Integrity
- Center for Vascular Research
- Center for Genome Engineering
- Center for Microbiome–Body–Brain Physiology
- Earth Science
- Interdisciplinary
- Center for Neuroscience Imaging Research (Neuro Technology Group)
- Center for Neuroscience Imaging Research (Cognitive and Computational Neuroscience Group)
- Center for Algorithmic and Robotized Synthesis
- Center for Nanomedicine
- Center for Biomolecular and Cellular Structure
- Center for 2D Quantum Heterostructures
- Institutes
- Korea Virus Research Institute
- News Center 뉴스 센터
- Career 인재초빙
- Living in Korea IBS School-UST
- IBS School 윤리경영


주메뉴
- About IBS
-
Research Centers
- Research Outcomes
- Mathematics
- Physics
- Center for Underground Physics
- Center for Theoretical Physics of the Universe (Particle Theory and Cosmology Group)
- Center for Theoretical Physics of the Universe (Cosmology, Gravity and Astroparticle Physics Group)
- Dark Matter Axion Group
- Center for Artificial Low Dimensional Electronic Systems
- Center for Quantum Nanoscience
- Center for Exotic Nuclear Studies
- Center for Van der Waals Quantum Solids
- Center for Relativistic Laser Science
- Center for Trapped Ion Quantum Science
- Chemistry
- Life Sciences
- Center for Memory and Glioscience (Cognitive Glioscience Group)
- Center for Memory and Glioscience (Learning and Memory Group)
- Center for Synaptic Brain Dysfunctions
- Center for RNA Research
- Center for Genomic Integrity
- Center for Vascular Research
- Center for Genome Engineering
- Center for Microbiome–Body–Brain Physiology
- Earth Science
- Interdisciplinary
- Center for Neuroscience Imaging Research (Neuro Technology Group)
- Center for Neuroscience Imaging Research (Cognitive and Computational Neuroscience Group)
- Center for Algorithmic and Robotized Synthesis
- Center for Nanomedicine
- Center for Biomolecular and Cellular Structure
- Center for 2D Quantum Heterostructures
- Institutes
- Korea Virus Research Institute
- News Center
- Career
- Living in Korea
- IBS School
News Center
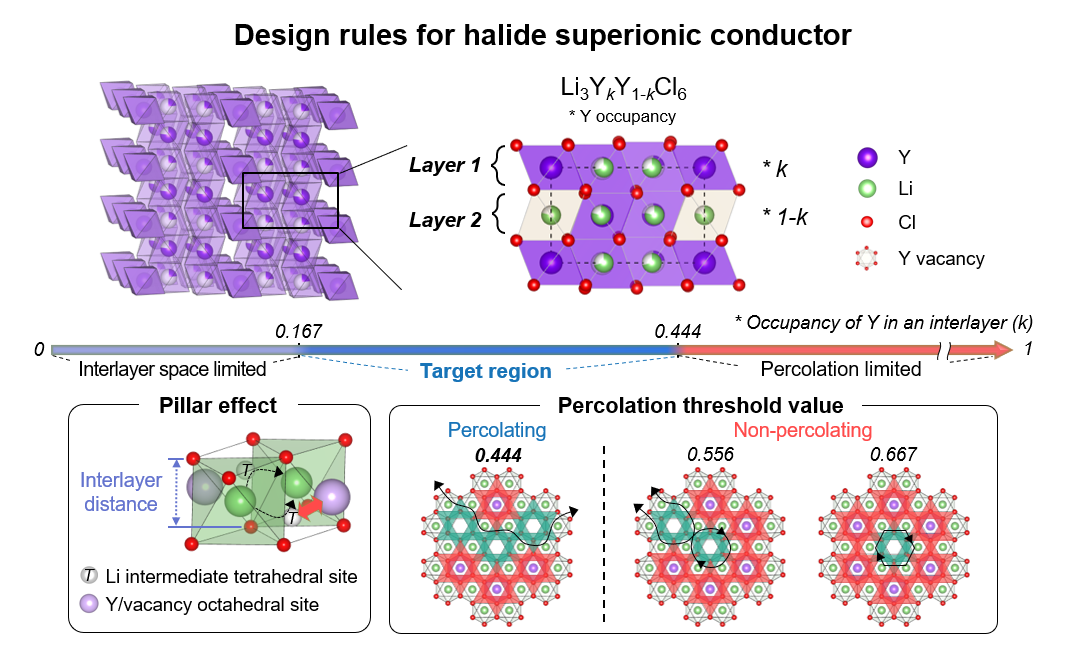
New designs for solid-state electrolytes may soon revolutionize the battery industry- Scientists achieve monumental improvements in lithium-metal-chloride solid-state electrolytes - Researchers led by Professor KANG Kisuk of the Center for Nanoparticle Research within the Institute for Basic Science (IBS), have announced a major breakthrough in the field of next-generation solid-state batteries. It is believed that their new findings will enable the creation of batteries based on a novel chloride-based solid electrolyte that exhibits exceptional ionic conductivity. A pressing concern with current commercial batteries their reliance on liquid electrolytes, which leads to flammability and explosion risks. Therefore, the development of non-combustible solid electrolytes is of paramount importance for advancing solid-state battery technology. As the world gears up to regulate internal combustion engine vehicles and expand the use of electric vehicles in the ongoing global shift toward sustainable transportation, research into the core components of secondary batteries, particularly solid-state batteries, has gained significant momentum. To make solid-state batteries practical for everyday use, it is crucial to develop materials with high ionic conductivity, robust chemical and electrochemical stability, and mechanical flexibility. While previous research successfully led to sulfide and oxide-based solid electrolytes with high ionic conductivity, none of these materials fully met all these essential requirements. In the past, scientists have also explored chloride-based solid electrolytes, known for their superior ionic conductivity, mechanical flexibility, and stability at high voltages. These properties led some to speculate that chloride-based batteries are the most likely candidates for solid-state batteries. However, these hopes quickly died out, as the chloride batteries were considered impractical due to their heavy reliance on expensive rare earth metals, including yttrium, scandium, and lanthanide elements, as secondary components. To address these concerns, the IBS research team looked at the distribution of metal ions in chloride electrolytes. They believed the reason trigonal chloride electrolytes can achieve low ionic conductivity is based on the variation of metal ion arrangements within the structure. They first tested this theory on lithium yttrium chloride, a common lithium metal chloride compound. When the metal ions were positioned near the pathway of lithium ions, electrostatic forces caused obstruction in their movement. Conversely, if the metal ion occupancy was too low, the path for lithium ions became too narrow, impeding their mobility. Building on these insights, the research team introduced strategies to design electrolytes in a way that mitigates these conflicting factors, ultimately leading to the successful development of a solid electrolyte with high ionic conductivity. The group went further to successfully demonstrate this strategy by creating a lithium-metal-chloride solid-state battery based on zirconium, which is far cheaper than the variants that employ rare earth metals. This was the first instance where the significance of the metal ions arrangement on a material’s ionic conductivity was demonstrated. This research brings to light the often-overlooked role of metal ion distribution in the ionic conductivity of chloride-based solid electrolytes. It is expected that the IBS Center’s research will pave the way for the development of various chloride-based solid electrolytes and further drive the commercialization of solid-state batteries, promising improved affordability and safety in energy storage. Corresponding author KANG Kisuk states, “This newly discovered chloride-based solid electrolyte is poised to transcend the limitations of conventional sulfide and oxide-based solid electrolytes, bringing us one step closer to the widespread adoption of solid-state batteries.” This research was published on November 3, 2023, in Science, which is one of the world's most prestigious scientific journals.
Notes for editors
- References
- Media Contact
- About the Institute for Basic Science (IBS)
|
| Next | |
|---|---|
| before |
- Content Manager
- Public Relations Team : Suh, William Insang 042-878-8137
- Last Update 2023-11-28 14:20