주메뉴
- About IBS 연구원소개
-
Research Centers
연구단소개
- Research Outcomes
- Mathematics
- Physics
- Center for Underground Physics
- Center for Theoretical Physics of the Universe (Particle Theory and Cosmology Group)
- Center for Theoretical Physics of the Universe (Cosmology, Gravity and Astroparticle Physics Group)
- Dark Matter Axion Group
- Center for Artificial Low Dimensional Electronic Systems
- Center for Quantum Nanoscience
- Center for Exotic Nuclear Studies
- Center for Van der Waals Quantum Solids
- Center for Relativistic Laser Science
- Center for Trapped Ion Quantum Science
- Chemistry
- Life Sciences
- Center for Memory and Glioscience (Cognitive Glioscience Group)
- Center for Memory and Glioscience (Learning and Memory Group)
- Center for Synaptic Brain Dysfunctions
- Center for RNA Research
- Center for Genomic Integrity
- Center for Vascular Research
- Center for Genome Engineering
- Center for Microbiome–Body–Brain Physiology
- Earth Science
- Interdisciplinary
- Center for Neuroscience Imaging Research (Neuro Technology Group)
- Center for Neuroscience Imaging Research (Cognitive and Computational Neuroscience Group)
- Center for Algorithmic and Robotized Synthesis
- Center for Nanomedicine
- Center for Biomolecular and Cellular Structure
- Center for 2D Quantum Heterostructures
- Institutes
- Korea Virus Research Institute
- News Center 뉴스 센터
- Career 인재초빙
- Living in Korea IBS School-UST
- IBS School 윤리경영


주메뉴
- About IBS
-
Research Centers
- Research Outcomes
- Mathematics
- Physics
- Center for Underground Physics
- Center for Theoretical Physics of the Universe (Particle Theory and Cosmology Group)
- Center for Theoretical Physics of the Universe (Cosmology, Gravity and Astroparticle Physics Group)
- Dark Matter Axion Group
- Center for Artificial Low Dimensional Electronic Systems
- Center for Quantum Nanoscience
- Center for Exotic Nuclear Studies
- Center for Van der Waals Quantum Solids
- Center for Relativistic Laser Science
- Center for Trapped Ion Quantum Science
- Chemistry
- Life Sciences
- Center for Memory and Glioscience (Cognitive Glioscience Group)
- Center for Memory and Glioscience (Learning and Memory Group)
- Center for Synaptic Brain Dysfunctions
- Center for RNA Research
- Center for Genomic Integrity
- Center for Vascular Research
- Center for Genome Engineering
- Center for Microbiome–Body–Brain Physiology
- Earth Science
- Interdisciplinary
- Center for Neuroscience Imaging Research (Neuro Technology Group)
- Center for Neuroscience Imaging Research (Cognitive and Computational Neuroscience Group)
- Center for Algorithmic and Robotized Synthesis
- Center for Nanomedicine
- Center for Biomolecular and Cellular Structure
- Center for 2D Quantum Heterostructures
- Institutes
- Korea Virus Research Institute
- News Center
- Career
- Living in Korea
- IBS School
News Center
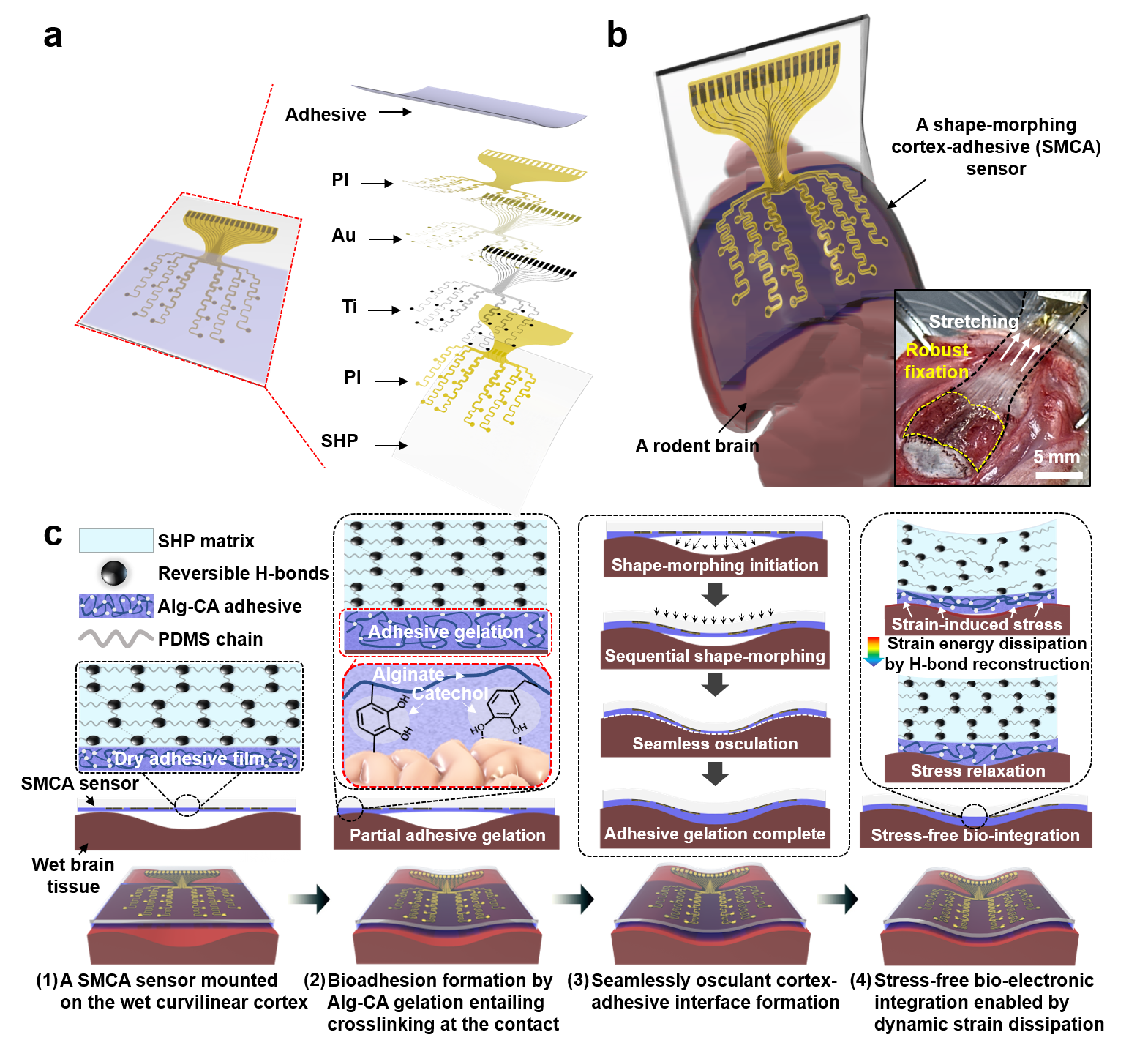
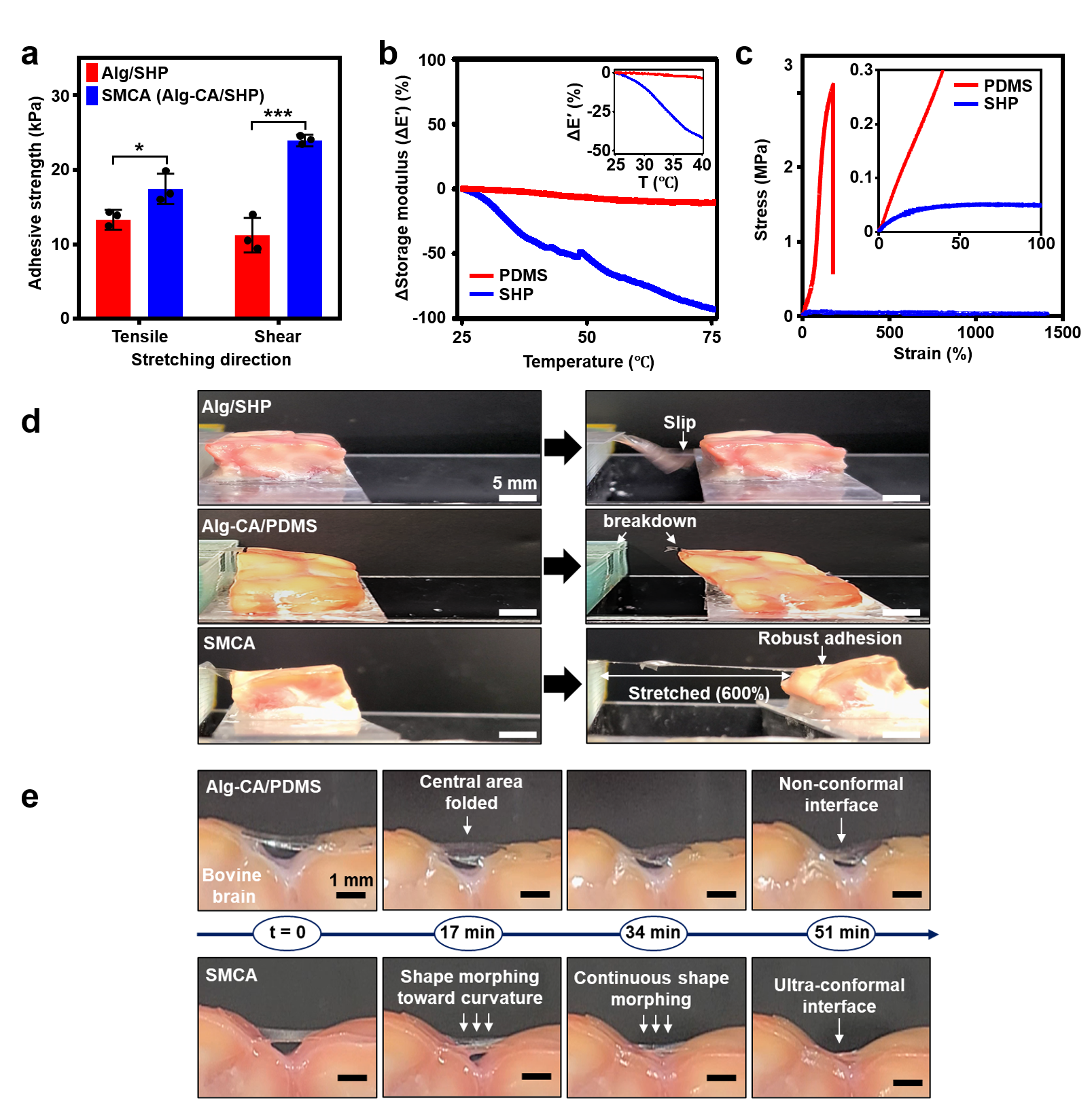
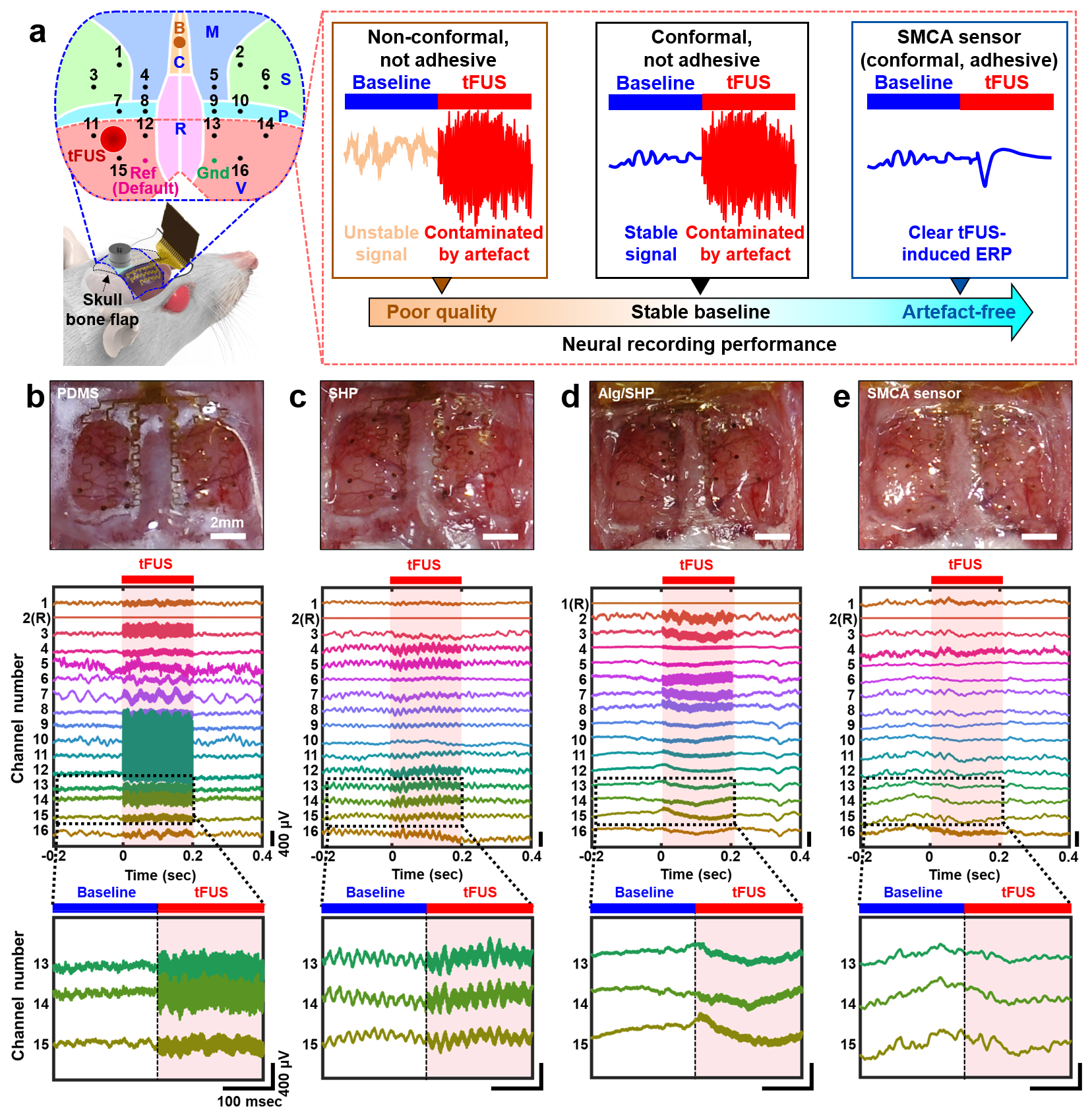
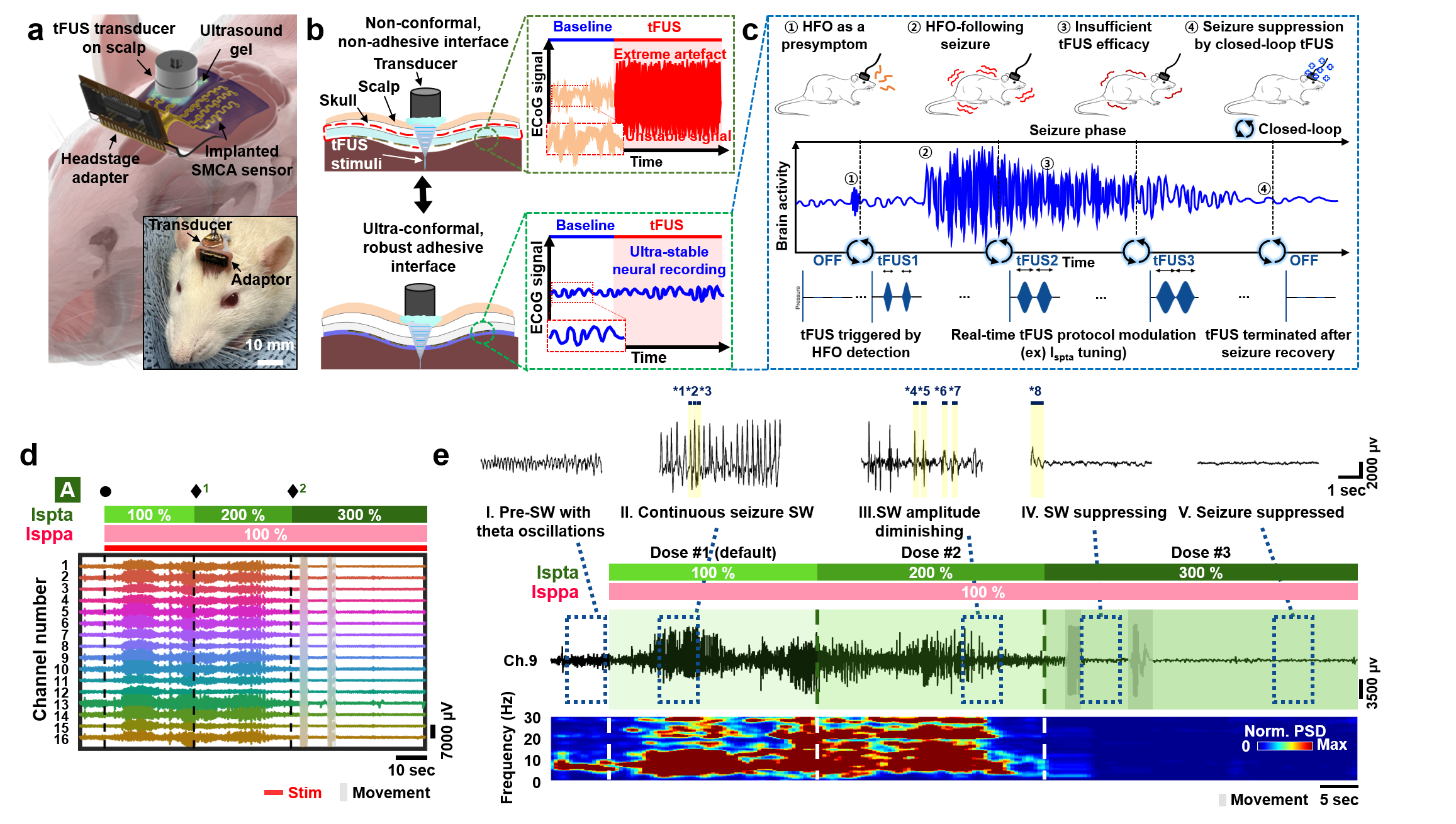
Adhesive Cortical Device Enables Artifact-Free Neuromodulation for Closed-loop Epilepsy Treatment- A shape-morphing cortex-adhesive sensor realizes personalized focused ultrasound neurostimulation through high-fidelity electrocorticography - A team of researchers led by Professor SON Donghee and Professor SHIN Mikyung from the Center for Neuroscience Imaging Research (CNIR) within the Institute for Basic Science (IBS), along with Dr. KIM Hyungmin from the Bionics Research Center at the Korea Institute of Science and Technology (KIST), has developed a groundbreaking soft cortical device that could revolutionize the treatment of epilepsy and other neurological disorders. Epilepsy, a neurological disorder affecting over 65 million people worldwide, is characterized by pathological electrical hyperactivity in the brain resulting in seizures. Notably, approximately 20-30% of all patients are diagnosed with intractable epilepsy, which does not respond to standard medications. Surgical resection of lesions remains a treatment option for these patients, but it presents challenges due to the complexity and risks involved in the procedure. As a less invasive alternative treatment, the concept of neuromodulation has been proposed, which involves directly stimulating lesioned tissue with mechanical, electromagnetic, or optical energy to suppress brain hyperexcitability. One promising approach is transcranial focused ultrasound (tFUS) neurostimulation, a non-invasive method that stimulates the brain with high precision without causing permanent damage. For tFUS to be effective in treating epilepsy, it must be paired with a system that can continuously monitor brain activity and adjust the treatment in real-time. However, existing cortex-interfacing devices face challenges due to their high stiffness and low shape adaptability, which makes it difficult for them to conform to the convoluted surface of the brain, resulting in poor tissue-device interfaces. Their low adhesion to the brain surface also means they struggle to provide accurate brain signals during ultrasound stimulation, due to the interference caused by the mechanical pressure waves. To address this challenge, the research team developed the Shape-Morphing Cortical-Adhesive (SMCA) sensor, a soft, flexible device that adheres closely to the brain’s surface, ensuring stable and accurate monitoring of brain activity even during tFUS stimulation. The SMCA sensor is composed of a unique combination of materials. It features a layer of catechol-conjugated alginate hydrogel that quickly bonds with brain tissue, providing strong adhesion and reducing the risk of movement or detachment. Additionally, the device’s substrate is made of a self-healing polymer that softens and conforms to the brain’s curved surface at body temperature, ensuring a snug fit and minimizing the risk of signal artifacts. The team tested the SMCA sensor both ex vivo (outside the body) and in vivo (inside the body), comparing its performance to that of existing devices without adhesive or shape-morphing properties. In experiments with a rat model of epilepsy, the SMCA sensor successfully recorded brain activity during tFUS without interference, enabling the real-time monitoring necessary for effective treatment. Using this innovative sensor, the researchers implemented a closed-loop seizure control system. This system uses the SMCA sensor to detect early signs of a seizure and automatically adjusts the tFUS treatment in response. The system successfully suppressed seizures in real-time, demonstrating the potential for personalized, adaptive epilepsy treatment. Professor SON Donghee stated, “Through our study on the brain-adhesive soft bioelectronics platform, we have overcome a major challenge in the field of brain interfaces by achieving high-quality electrocorticography coupled with focused ultrasound stimulation without artifact interference.” He explained the significance of this research and outlined future plans by adding, “We expect our technology to become a cornerstone of a next-generation biomedical platform that enables precise diagnosis and personalized therapy for intractable neurological disorders. Following this study, we will advance the SMCA sensor platform by improving the shape-morphing and cortex-adhesive functionalities, developing highly integrated microelectrodes, and implementing a high-order closed-loop operational algorithm.” Dr. Hyungmin KIM stated, “We achieved early detection of seizure activity via ECoG, enabling the prevention of seizures. Additionally, we implemented real-time feedback on the effects of ultrasound stimulation, which allowed for the application of personalized stimulation protocols. Looking ahead, we anticipate that the development of electrodes with more channels, as well as multi-channel ultrasound transducers, will facilitate precise mapping of seizure sources and targeted intervention, ultimately enhancing the efficacy and safety of this approach in clinical applications.” This research was conducted in collaboration with colleagues from Sungkyunkwan University (SKKU) and the Korea Institute of Science and Technology (KIST). The findings were published in Nature Electronics on September 11, 2024.
Schematic illustrations of sequential brain-interfacing steps of the SMCA sensor for explaining the tissue-adhesive shape-morphing mechanism.
Notes for editors
- References
- Media Contact
- About the Institute for Basic Science (IBS)
|
| Next | |
|---|---|
| before |
- Content Manager
- Public Relations Team : Suh, William Insang 042-878-8137
- Last Update 2023-11-28 14:20